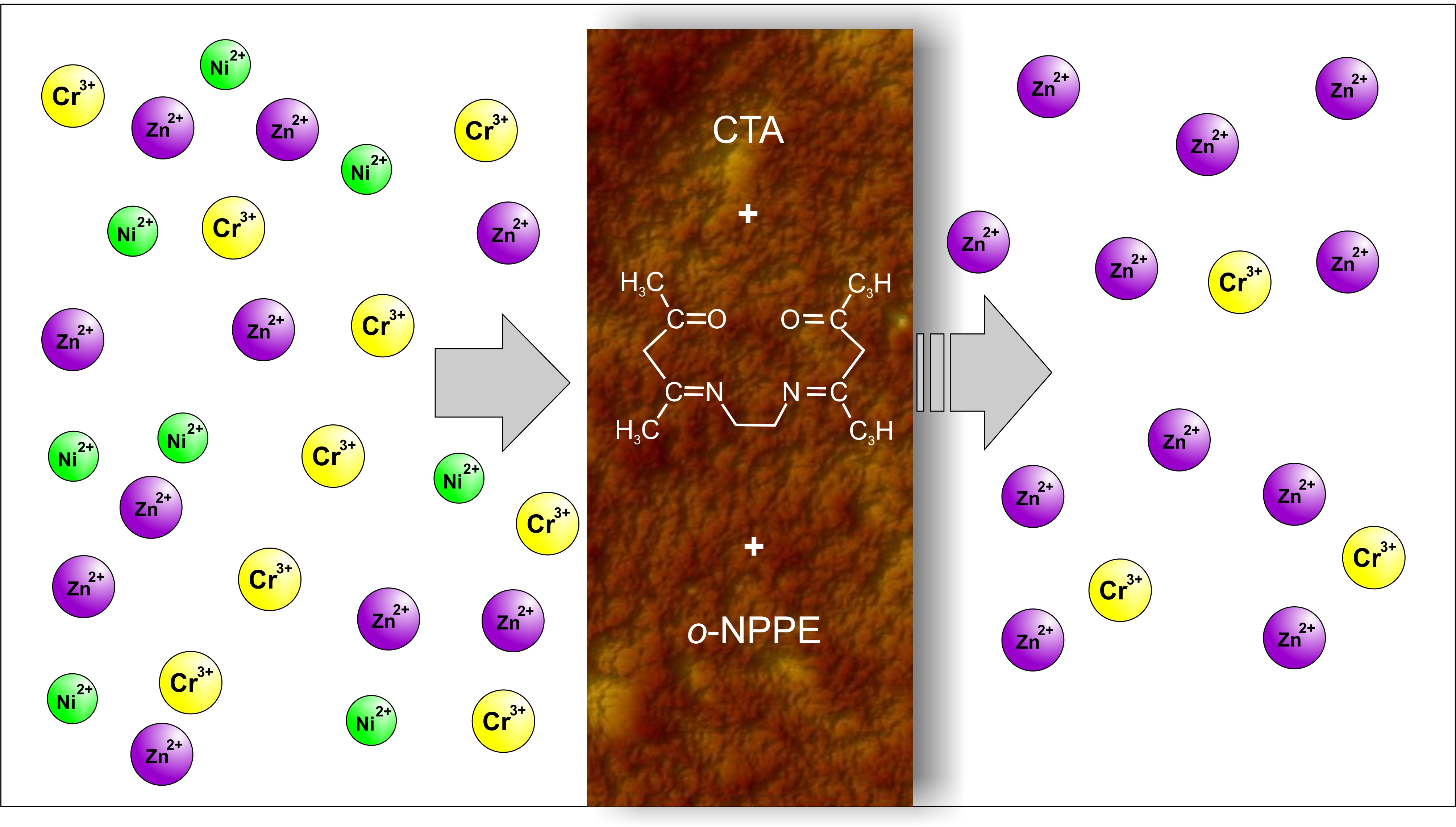
Membranes | Free Full-Text | Separation of Zn(II), Cr(III), and Ni(II) Ions Using the Polymer Inclusion Membranes Containing Acetylacetone Derivative as the Carrier

Warmup 2. Balance this redox reaction in acid: + Fe 2+ + Cr 2 O 7 2- Fe 3+ + Cr 3+ Ni e¯ Ni E° = V Zn e¯ Zn E° = V 1. Write. - ppt download

Figure 2 from Ti-Cu-Ni(Fe,Cr,Co)-Sn-Ta(Nb) Alloys with Potential for Biomedical Applications | Semantic Scholar

Structure and Bonding of the Transition-Metal Carbonyl Complexes M(CO)5L (M = Cr, Mo, W) and M(CO)3L (M = Ni, Pd, Pt; L = CO, SiO, CS, N2, NO+, CN-, NC-, HCCH, CCH2,
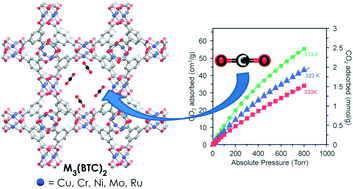
Investigation of the synthesis, activation, and isosteric heats of CO2 adsorption of the isostructural series of metal–organic frameworks M3(BTC)2 (M = Cr, Fe, Ni, Cu, Mo, Ru) - Dalton Transactions (RSC Publishing)
![i) Write the IUPAC name of the complex [ Cr (NH3)4Cl2 ] Cl .(ii) What type of isomerism is exhibited by the complex [ Co (en)3 ]^3 + ?( en = ethane - i) Write the IUPAC name of the complex [ Cr (NH3)4Cl2 ] Cl .(ii) What type of isomerism is exhibited by the complex [ Co (en)3 ]^3 + ?( en = ethane -](https://dwes9vv9u0550.cloudfront.net/images/3015405/99fcb6fb-74fb-4941-944b-9d7ecaa533c9.jpg)
i) Write the IUPAC name of the complex [ Cr (NH3)4Cl2 ] Cl .(ii) What type of isomerism is exhibited by the complex [ Co (en)3 ]^3 + ?( en = ethane -

Pb^(+2) + 2e^(-) rarr Pb(s), E^(@) = -0.13V Sn^(+2) + 2e^(-) rarr Sn(s), E^(@) = - 0.16V Ni^(+2) + 2e^(-) rarr Ni(s), E^(@) = -0.25V Cr^(+3) + 3e^(-) rarr Cr(s), E^(@) = -

For the redox reaction `Cr_(2)O_(7)^(-2)+H^(+)+Ni rarr Cr^(3)+Ni^(2+)+H_(2)O` The correct coefficent - YouTube

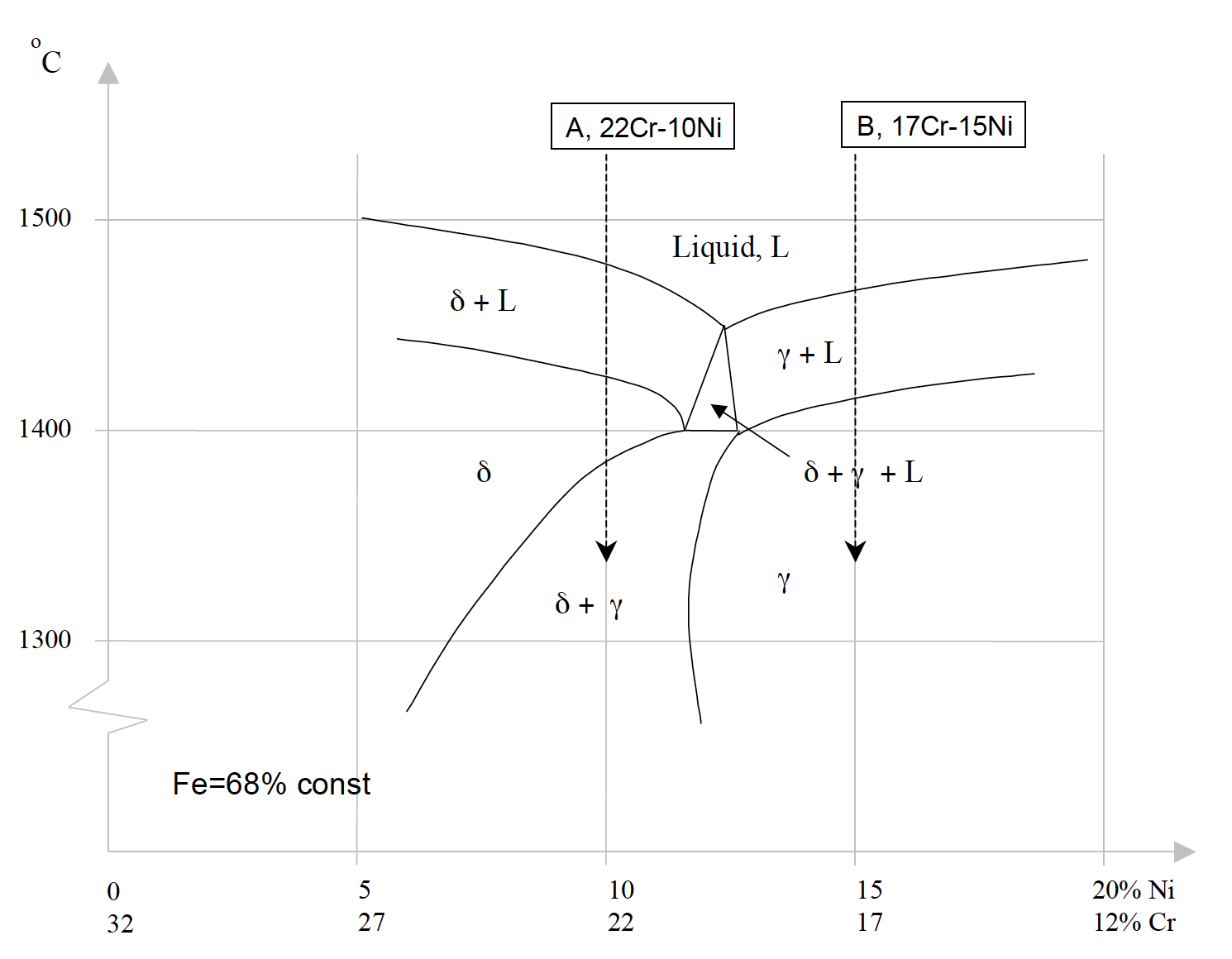
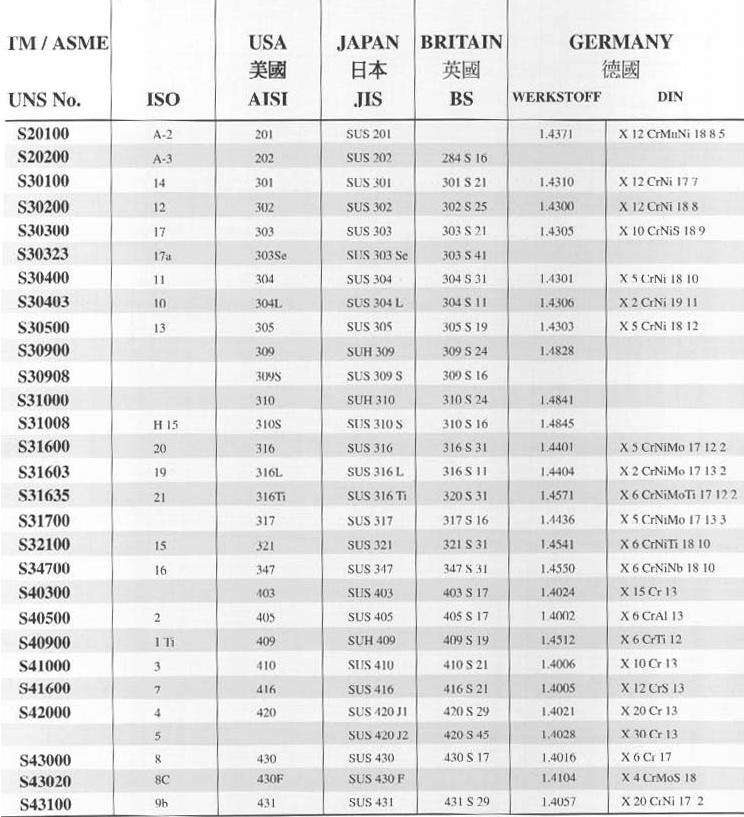
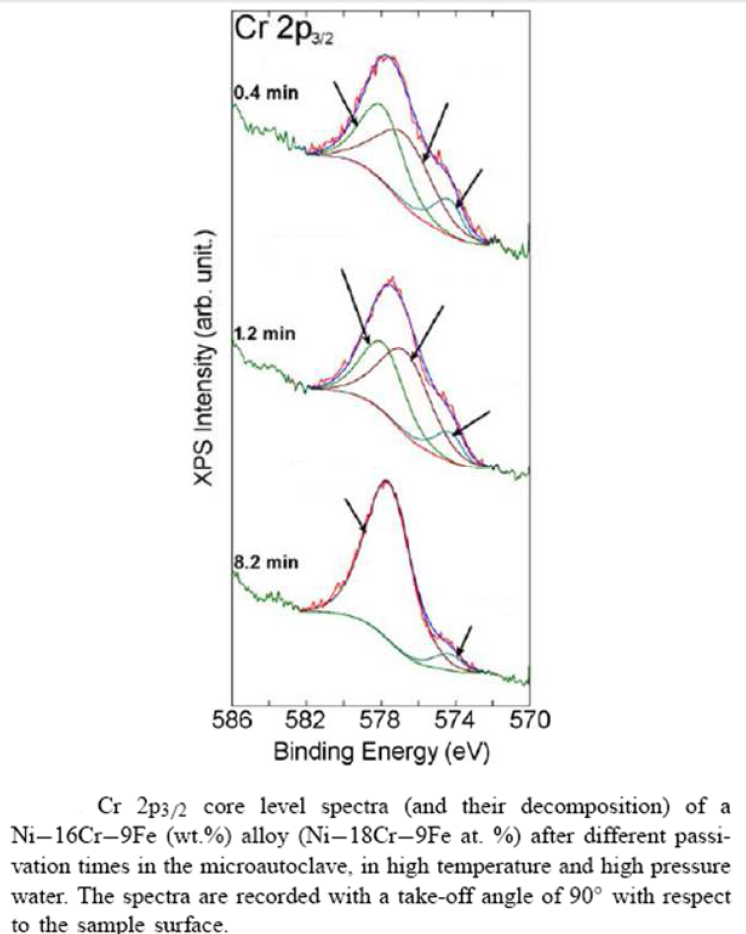
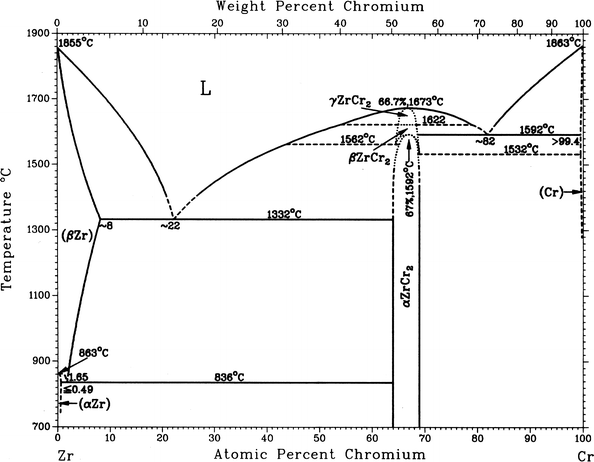




![Vertical Section of the Fe-Cr-Ni Phase Diagram at 70% Fe [2] | Download Scientific Diagram Vertical Section of the Fe-Cr-Ni Phase Diagram at 70% Fe [2] | Download Scientific Diagram](https://www.researchgate.net/publication/328585108/figure/fig1/AS:687292536602628@1540874718643/Vertical-Section-of-the-Fe-Cr-Ni-Phase-Diagram-at-70-Fe-2.png)


