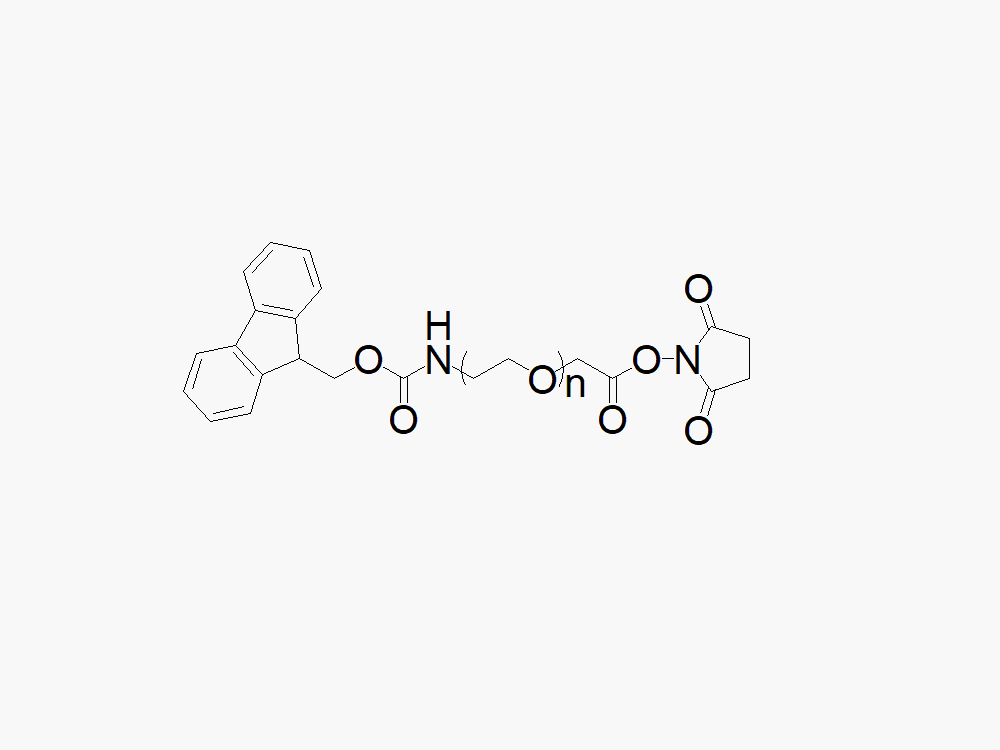
Cas(82911-69-1), Fmoc N-hydroxysuccinimide ester-Aladdin, 9-Fluorenylmethyl succinimidyl carbonate; N-(9-Fluorenylmethoxycarbonyloxy) succinimide; 9-Fluorenylmethyl N-succinimidyl carbonate; Carbonic Acid 9-Fluorenylmethyl N-Succinimidyl Ester; N-(Fmoc-ox

Problems in the synthesis of cyclic peptides through use of the Dmab protecting group - Journal of the Chemical Society, Perkin Transactions 1 (RSC Publishing) DOI:10.1039/B001694M

Solid-phase synthesis of compound RZ1. (i) Fmoc-EDA·HCl, DIEA, CH2Cl2,... | Download Scientific Diagram

Attempts Towards the Bucky-amino Acid Acylation of the Phospho-cytidine-phospho-adenosine (pdCpA) Subunit | Strom | The All Results Journals: Nano

Arylboronate Ester Protected Amino Acids as Orthogonal Building Blocks for Fmoc Solid‐Phase Peptide Synthesis - Liu - 2017 - European Journal of Organic Chemistry - Wiley Online Library
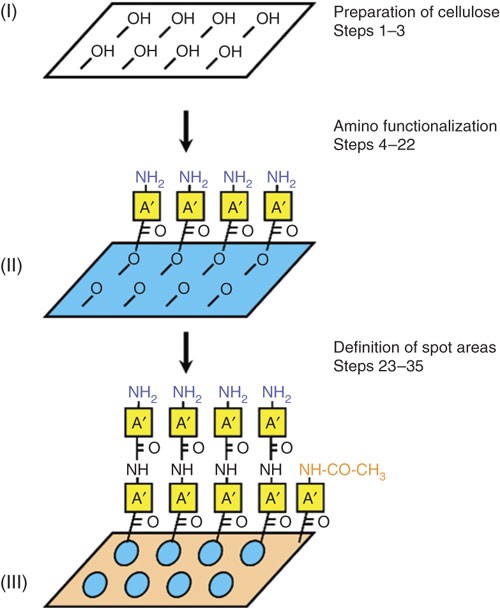
Peptide arrays on cellulose support: SPOT synthesis, a time and cost efficient method for synthesis of large numbers of peptides in a parallel and addressable fashion | Nature Protocols

Total Synthesis of Uniflorine A, Casuarine, Australine, 3-epi-Australine, and 3,7-Di-epi-australine from a Common Precursor | The Journal of Organic Chemistry
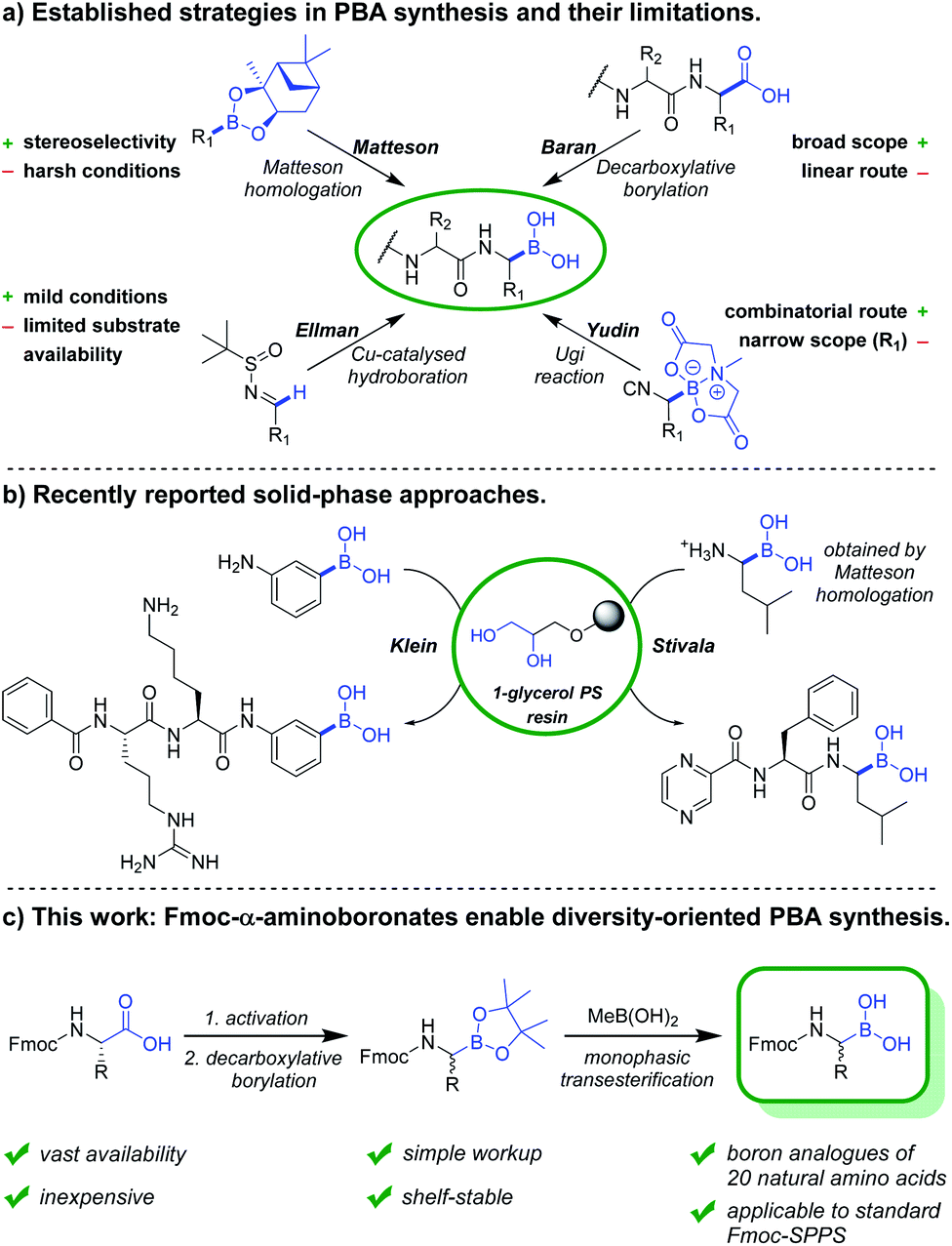
Diversity-oriented synthesis of peptide-boronic acids by a versatile building-block approach - Chemical Science (RSC Publishing) DOI:10.1039/D0SC03999C