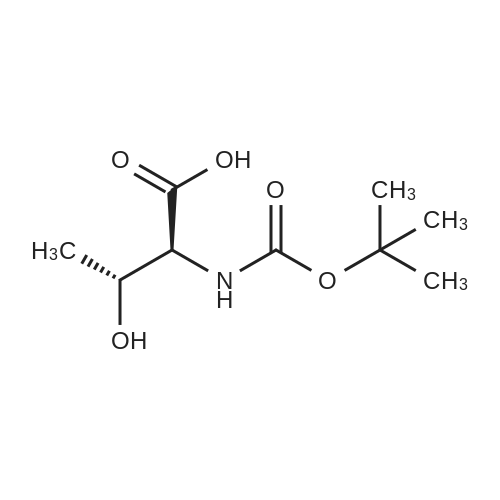
Convenient Synthesis of N‐Terminal Tfm‐Dipeptides from Unprotected Enantiopure α‐Tfm‐Proline and α‐Tfm‐Alanine - Chaume - 2009 - European Journal of Organic Chemistry - Wiley Online Library

Structural and functional analysis of EntV reveals a 12 amino acid fragment protective against fungal infections | Nature Communications

Molecules | Free Full-Text | Synthesis of Cyclic Peptides in SPPS with Npb-OH Photolabile Protecting Group
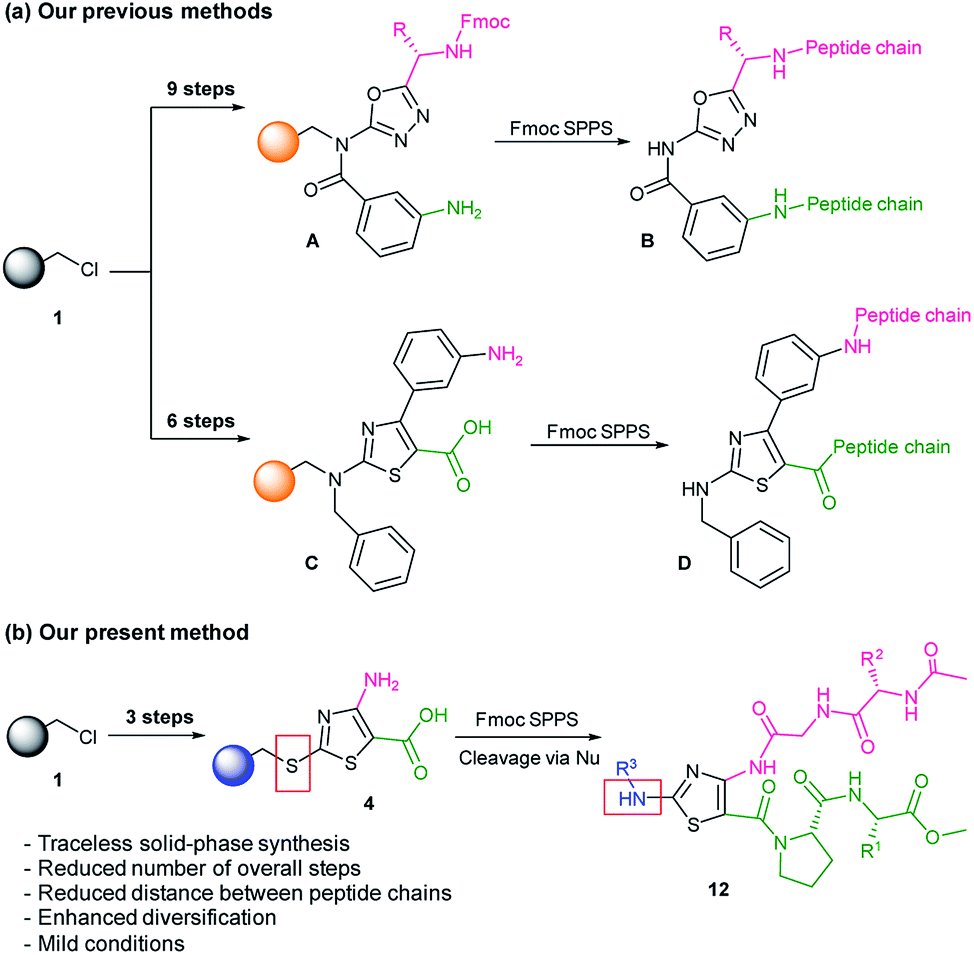
Traceless solid-phase synthesis and β-turn propensity of 1,3-thiazole-based peptidomimetics - RSC Advances (RSC Publishing) DOI:10.1039/D0RA10127C

Proteomic data and structure analysis combined reveal interplay of structural rigidity and flexibility on selectivity of cysteine cathepsins | Communications Biology
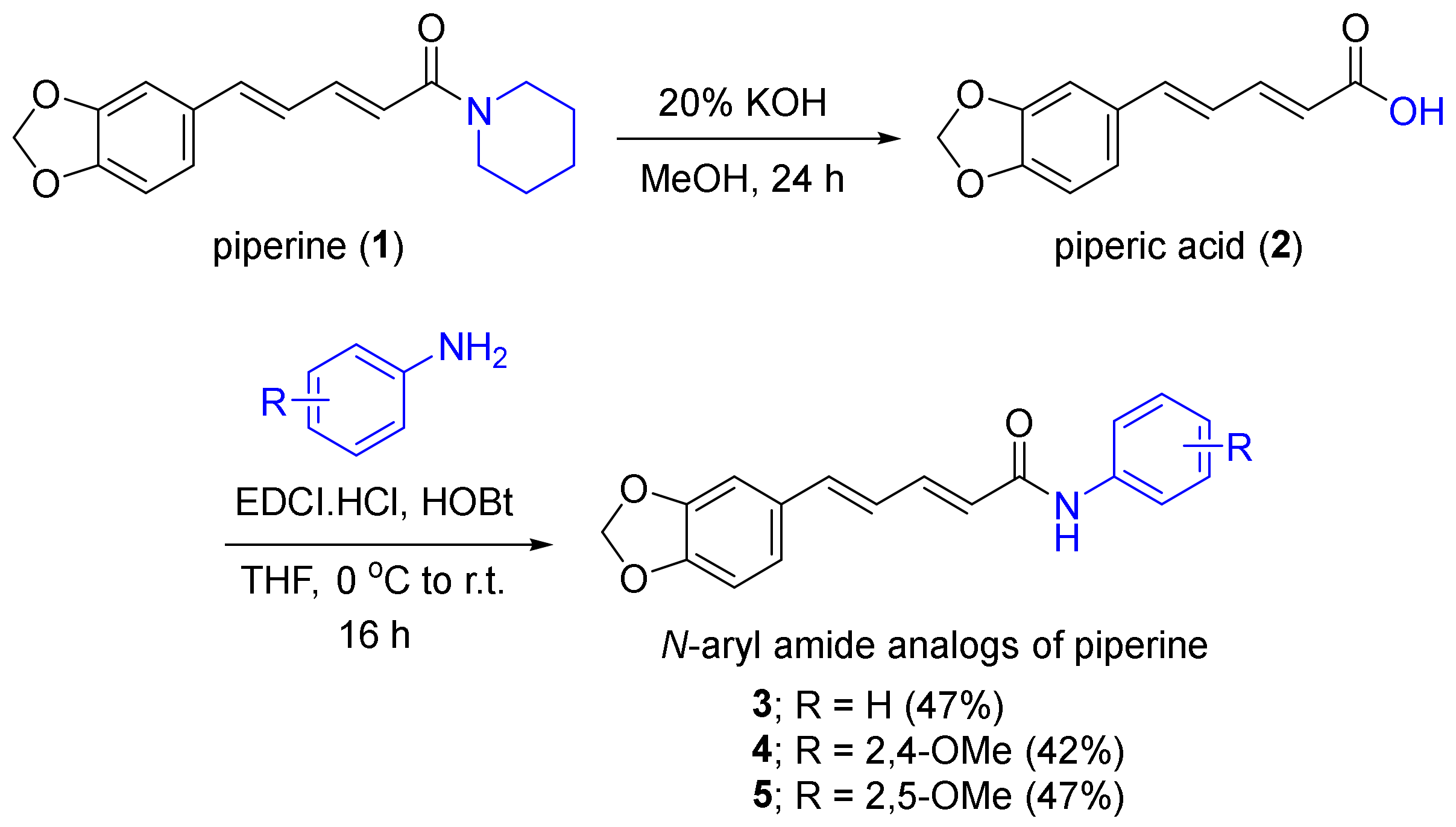
Molecules | Free Full-Text | Semi-Synthesis of N-Aryl Amide Analogs of Piperine from Piper nigrum and Evaluation of Their Antitrypanosomal, Antimalarial, and Anti-SARS-CoV-2 Main Protease Activities

PDF) 4-Aminophthalimide Amino Acids as Small and Environment-Sensitive Fluorescent Probes for Transmembrane Peptides

Discovery of Narrow Spectrum Kinase Inhibitors: New Therapeutic Agents for the Treatment of COPD and Steroid-Resistant Asthma | Journal of Medicinal Chemistry

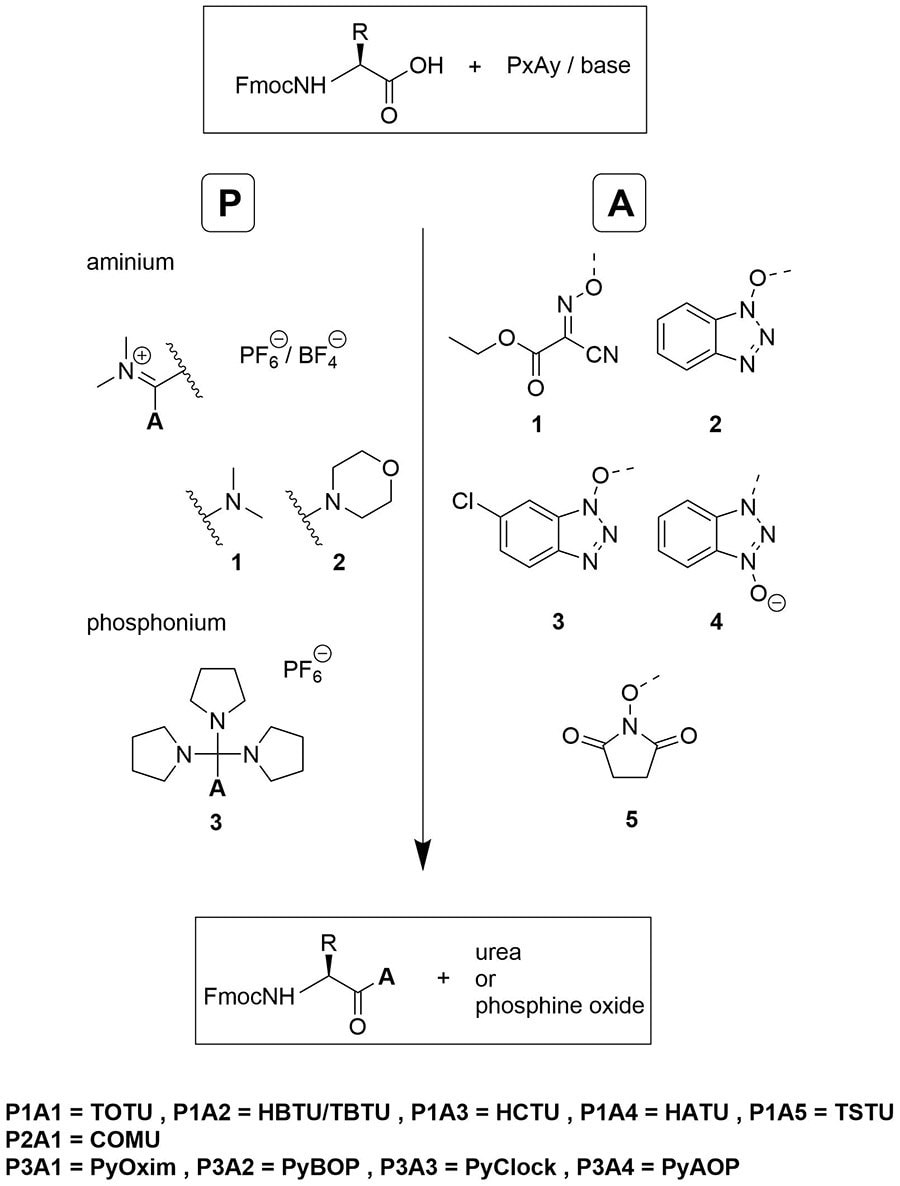
A Novel Family of Onium Salts Based Upon Isonitroso Meldrum's Acid Proves Useful as Peptide Coupling Reagents - El‐Faham - 2010 - European Journal of Organic Chemistry - Wiley Online Library

Visible Light‐Mediated Functionalization of Selenocystine‐Containing Peptides - Lapcinska - 2021 - Advanced Synthesis & Catalysis - Wiley Online Library